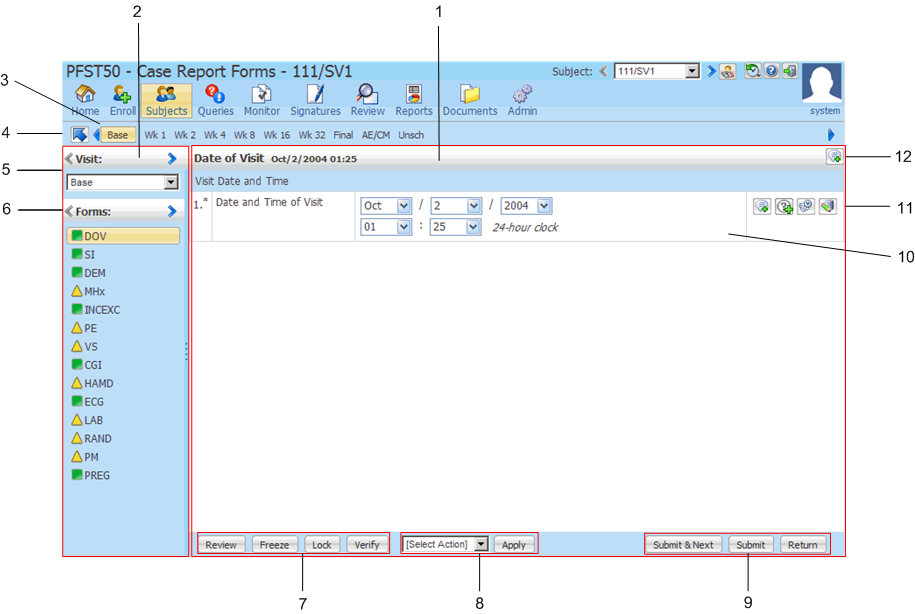
40% of respondents deemed Medidata their top pick for Phase I/II, Phase IIb/III, and Phase IV post-marketing trials. )/Img/medidata.jpg)
In the same survey mentioned above, Medidata’s Rave EDC was the runaway first-choice preference for all trial types. Medidata’s solutions scale to meet the demands of any size-Rave EDC can scale from a few sites and healthy volunteers to mega-trials with thousands of sites and tens of thousands of patients. Medidata’s Rave EDC easily allows for scalability as a sponsor or CRO moves into Phase II, Phase III, and Phase IV trials, involving more patients and more data. Subject data can be migrated from one form or protocol version to a new version.įor adaptive trials, Rave EDC allows granular control over extensive study design changes by configuring and testing the changes before deployment (with a full audit trail). Protocol amendments and incremental changes are implemented with no system downtime. In a recent Industry Standard Research survey, 42% of respondents said Medidata was their most preferred EDC provider for Phase I and II trials (the highest rated provider) 1. In fact, this service is a fit for about 75% of the protocols we evaluate, and most study startups can be accomplished in less than two weeks. Our customers have found that the utilization of standard forms from a predefined library drives faster study build timelines, simplifies data collection, reduces data clean and reconciliation efforts, and results in shorter database lock timelines. The pre-configured library contains: common Phase I forms, fields, edit checks, and custom functions basic visit structure and dynamics and lab administration. We provide a Phase I-specific, pre-configured, and accelerated EDC implementation service that improves quality and reduces build times because it’s pre-validated and doesn’t require additional customization and testing. Medidata’s site-based pricing model means we can price appropriately for the budget of smaller Phase I clinical trials. Medidata delivers tailored, flexible, and cost-effective support for sponsors and CROs executing Phase I clinical trials through our unparalleled experience and best practices, conducting over 4,000 Phase I studies for 400 clients over more than 20 years. In this final blog, we will look at Medidata’s Rave EDC solution and how it can support your study needs-from early-stage trials through post-market surveillance. In the second blog, we discussed why an EDC system is so valuable and things to consider when choosing an EDC system provider. In the first blog in this series, we looked at the increased complexity of Phase I clinical trials and how an Electronic Data Capture (EDC) system can be hugely beneficial in this early stage. Katrina has grown and cultivated global partnerships at Medidata for over a decade, expanding the reach of the company’s transformative platform for clinical development, commercial, and real-world data. This blog was authored by Katrina Weigold, Vice President of Global Partners at Medidata. Proactive risk mitigation for research data quality issues to help prevent trial delays.Įmpowering a risk-based approach to source data verification by focusing on critical data.Ī cloud-based remote site access and monitoring solution. Quickly and accurately develop investigator grant budgets.Ī dynamic digital solution for risk assessment, monitoring, and mitigation.Ī powerful data & risk surveillance tool to improve data integrity & reduce trial risk. Unify your clinical research study data and financial management in one platform. Manage electronic trial master file content while maintaining readiness & compliance. Improve speed and efficiency for the oversight of studies with Rave CTMS (Clinical Trial Management System). Seamlessly generate, distribute, & manage clinical study files at the end of a study.ĭrive faster timelines across the trial lifecycle with centralized data & analytics.Ī streamlined, single digital endpoint adjudication system. RTSM (Randomization and Trial Supply Management)Įliminate double data entry and change orders for randomization and trial supply management.Īutomate collection, transmission and tracking of adverse event data. 
Simplify complex data investigation and cleaning across many data sources. Gain visibility and full control over your imaging data while simplifying processes for sites, sponsors, and core labs. The evolution in automated medical coding for Rave EDC. 
Eliminate complex, manual processes & achieve higher quality data for faster insights.Ĭapture and manage your patient, site and lab in the most advanced, robust and secure electronic data capture system.Įquip your clinical trial study with fast, accurate medical coding.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
